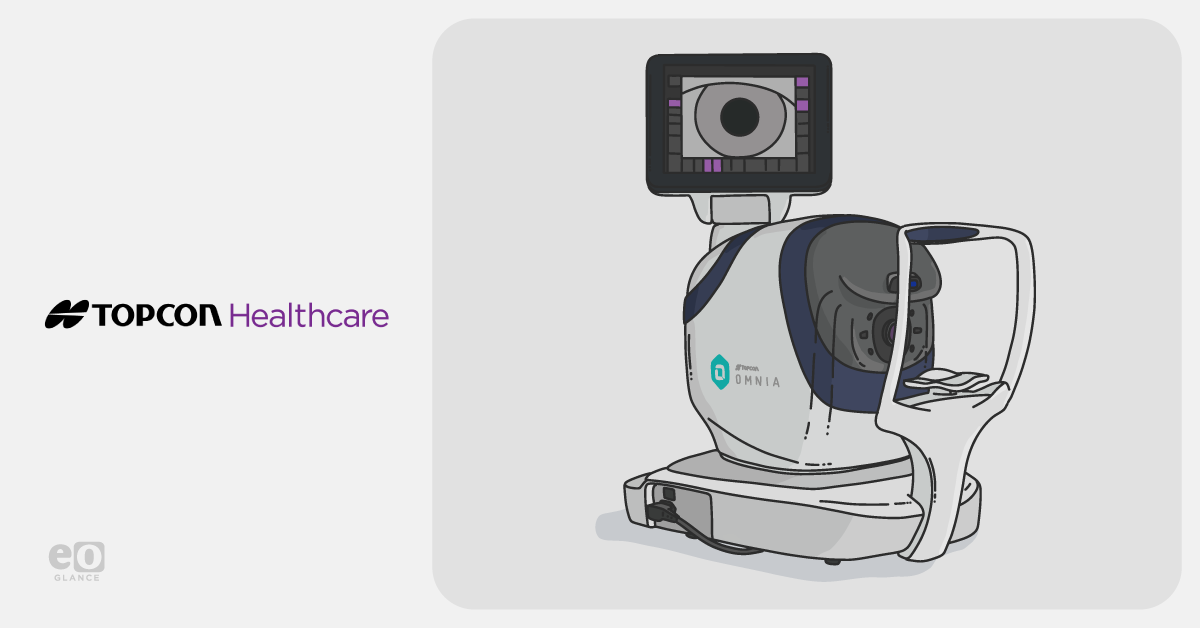
Topcon Healthcare, Inc. announced the simultaneous FDA 510(k) clearance—and U.S. commercial launch—of OMNIA, a fully automated…
Continue ReadingTag: FDA

QuantalX Receives FDA De Novo Clearance for Delphi-MD, Establishing a New Modality of Brain Function Assessment
“By unlocking direct insight into how the brain is functioning, Delphi-MD gives clinicians an essential piece…
Continue Reading
FDA clears Sora Neuroscience’s brain mapping software
Sora Neuroscience, a brain AI software company, announced it received FDA clearance for its Cirrus brain…
Continue Reading
FDA clears Hyperfine’s Optive AI software for brain imaging
Healthcare technology company Hyperfine announced that the FDA cleared its Optive AI software for its Swoop…
Continue Reading
GE HealthCare’s Revolutionary Brain MRI Scanner Gains FDA Clearance, Promises Faster Scans
GE HealthCare has received FDA 510(k) clearance for its SIGNA™ MAGNUS,…
Continue Reading
QuantalX Secures FDA Clearance for its breakthrough Delphi stimulator, advancing direct brain health diagnosis at the point of care
The Delphi-MD platform enables accurate, early detection of brain abnormalities across a wide range of conditions,…
Continue Reading