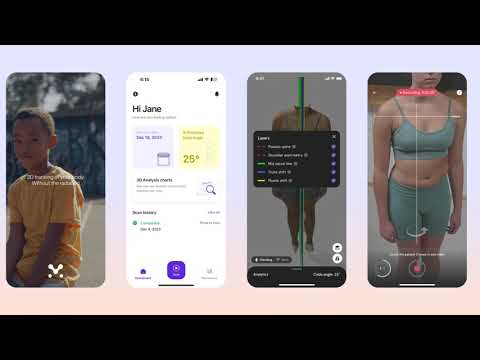
After receiving their Health Canada class 1 medical device clearance, Momentum Spine continues to gain traction…
Continue ReadingTag: 510k

DePuy Synthes receives FDA 510(k) clearance for TriALTIS Spine System
Johnson & Johnson MedTech has announced that DePuy Synthes, the orthopaedics company of Johnson & Johnson, has…
Continue Reading