Dive Brief: GE Healthcare has received 510(k) clearance for a tool designed to help clinicians assess…
Continue ReadingTag: FDA

Avicenna.AI lands FDA clearance for AI tool that detects cervical spine fracture
Medical imaging AI company Avicenna.AI has announced that it has received 510(k) clearance from the U.S. Food and…
Continue Reading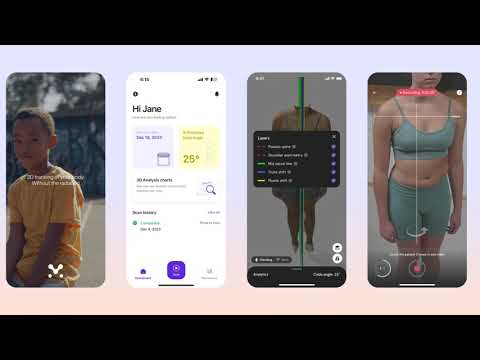
Momentum Health receives FDA 510K Clearance for Momentum Spine, a Pioneering AI-Enabled Mobile Application for Monitoring Scoliosis, Available for Download Now!
After receiving their Health Canada class 1 medical device clearance, Momentum Spine continues to gain traction…
Continue Reading
DePuy Synthes receives FDA 510(k) clearance for TriALTIS Spine System
Johnson & Johnson MedTech has announced that DePuy Synthes, the orthopaedics company of Johnson & Johnson, has…
Continue Reading
